

Desalination Processes
About 9% of the world population lack access to improved sources of drinking water and the need to potable water is constantly increasing due to the expanding world population. On the other hand, seas and oceans that contain 97% of the water on earth are a major alternative resource, yet unsuitable for human consumption without treatment since it contains typically 35000 parts per million (ppm) of salt (sodium chloride). Reverse osmosis (RO) is as a major technology for producing potable water from salt water using a salt-rejecting membrane under a pressure higher than the osmotic pressure in order to allow water to permeate through the membrane to obtain desalted water while rejecting the salt and other solutes. RO can be also used to produce potable water from inland brackish water whose salt content ranges from 500 ppm to 30000 ppm. The production of potable water from seawater by RO osmosis is more efficient compared to classical methods such as evaporation since it does not involve phase transition. However, the required pressure in RO is high in order to overcome the osmotic pressure difference, which contributes significantly to the cost of water desalination. Our research is focused on design and optimization of energy-efficient RO processes. Patent application for a new desalination process has been filed in Singapore.
Patent application for a new desalination process has been filed in Singapore (Application # 11201900705P).
In seawater desalination by reverse osmosis, commercial membranes (except some special-purpose ones) allow a maximum operating pressure of 69 bar. Hence, achieving overall potable water recoveries above 65% using Single-Stage-RO or Two-Stage-RO processes is not practically viable. The new process allows achieving high water recoveries (>70%) at pressures well below 69 bar with an electric energy consumption comparable to that of an SSRO.
Polymeric Gas Separation Membranes
Aromatic polyimides are an important group of polymer with outstanding thermal and chemical stability, and good mechanical properties, which make them very attractive as membrane material for membrane based gas separation. They are synthesized by the reaction of dianhydride and diaminemonomers, which govern selectivity towards separation of commercially important gas pairs such as O2/N2, CO2/CH4, H2/CH4, H2/N2 and propane/propylene. Hence, selection of an appropriate monomer pair is crucial for the preparation of membranes with high selectivity for a specific application.
Sorption induced volume swelling and plasticization is a serious concern limiting the use of these polymers as membrane material for gas separation involving CO2 and condensable hydrocarbons such as propylene and propane. Our research is focused on the study of the relationships between the chemical structure of the polyimides and their plasticization behavior.
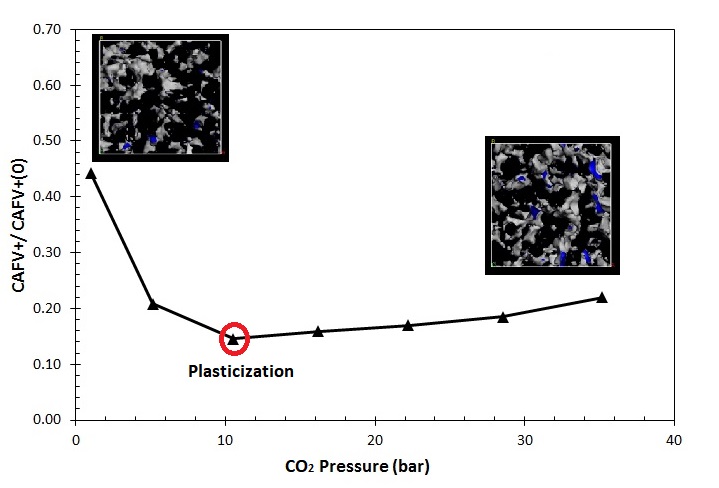
Predicted CO2-plasticization pressure of Matrimid®
MOFs for CO2 separation
Metal Organic Frameworks (MOFs) are novel candidates for gas separation processes and have significant advantages over conventional zeolites because of the favored interatomic interactions with gases, high surface area, free pore volume, high porosity, high flexibility and controllable surface properties and exhibit great potential in CO2 separation applications such as, CO2/CH4 and CO2/N2. as adsorbents or fillers in polymer-based mixed matrix membranes.
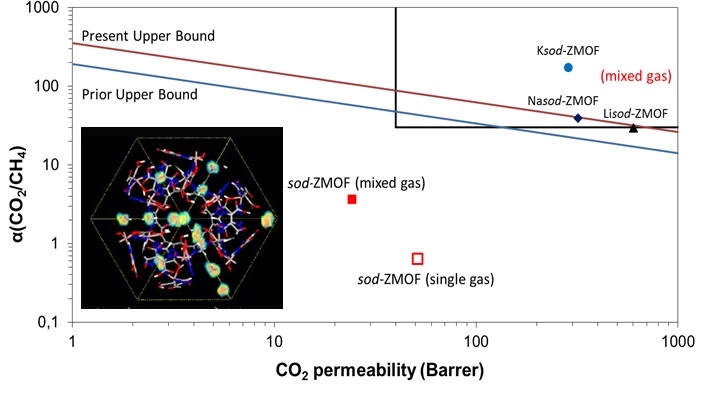
CO2/CH4 separation performance of ion-exchanged sod-ZMOF
Zeolites for Water Treatment
The chemical and structural features of zeolites make them very effective for the removal of toxic organics (VOCs, ammonia, ammonium) and metal ions (e.g. lead, nickel, zinc, cadmium, copper, chromium and/or cobalt), from municipal and industrial wastewaters.
Hyrdrophobic zeolites with high Si/Al ratio, such as ZSM-5, Zeolite Y, can adsorb organic vapors with molecules smaller than their pore size. The understanding of molecular interactions among organics, water and zeolite crystal is crucial to develop effective separation processes. Molecular simulation methods provide an opportunity to investigate these interactions at the atomistic scale.
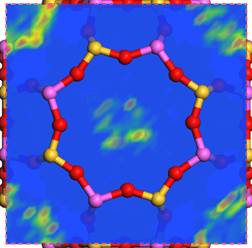
Silver ion distribution in a hydrated LTA alpha-cage
Surface Adsorption of Proteins
The surface adsorption of proteins onto materials is essential in various biomedical and biotechnological applications. An improved understanding of protein adsorption mechanism can therefore allow development of new materials for various applications, such as biocompatible materials for biomedical devices that are inserted in living bodies, biosensors that can detect the presence of target proteins, or purifiers that can absorb unwanted biological materials from a solution. Atomistic-level computational simulations can provide valuable insight on the details of interactions of individual amino acids with an inorganic surface.
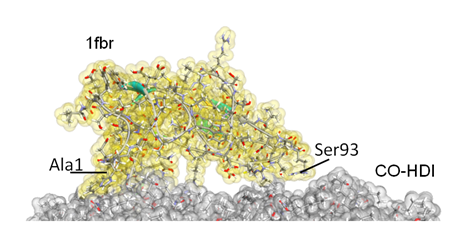
Fibronectin module (1fbr) adsorbed on a castor-oil-based polyurethane surface.

Prof. Göktuğ Ahunbay
Phone: +(90) 212-285 7217
Fax: +(90) 212-285 2925
E-mail: ahunbaym[at]itu.edu.tr
Faculty of Chemical and Metallurgical Engineering, Ayazaga Campus, Maslak 34469 Istanbul, TURKEY